AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
So enthalpy change of hydration and enthalpy change of lattice decrease. The solubility of group II hydroxides increase down the group as enthalpy change of solution is getting exothermic.Įnthalpy change of solution = Enthalpy change of hydration - Enthalpy change of lattice energy So enthalpy change of solution is getting endothermic. But the decrease in enthalpy change of hydration is more than the decrease in enthalpy change of lattice. Of solution of hydration of lattice energy The solubility of group II sulphates decreases down the group as the enthalpy change of solution is getting endothermic.Įnthalpy change = Enthalpy change - Enthalpy change So they decompose at low temperatures and in less time. The upper cation (smaller) can polarise the carbonate anion or nitrate anion more. As the size of the cation increases, its polarising power decreases. Chlorine and Argon do not react with oxygen.ĭown the group, the thermal stability of metal carbonates and metal nitrates of group II increases.Further oxidation of sulfur dioxide gives sulfur trioxide. Sulfur burns with a blue flame to form sulfur dioxide gas.Phosphorus burns with a yellow or white flame. Phosphorus reacts with oxygen to form phosphorus oxide.Silicon forms silicon dioxide by slowly reacting with oxygen.Powdered aluminium reacts with oxygen to form aluminium oxide.Magnesium reacts vigorously with oxygen to form magnesium oxide.Sodium vigorously reacts with oxygen to form a white solid of sodium oxide.Reactions of period 3 elements with oxygen: In general, the ionisation energies of period three elements increase across the period due to the increase in the positive nuclear charge. They are good insulators and have no electrical conductivity. Silicon, being a semiconductor, has a very low electrical conductivity. The electrical conductivity across period three increases from sodium to aluminium, as they have metallic bonding. The atomic radius of elements across period three decreases due to the increase in proton number and the same shielding. This recurrence of the same pattern is called periodicity. The patterns seen across period three are also seen across other periods. Group number tells us about the number of valence electrons of an element while the period number tells us about the number of shells occupied by those electrons. Elements in the same period have the same number of shells. Elements in the same group have the same number of valence electrons. The following diagram shows the structure of a carbon atom.Įlements are arranged in the periodic table in order of atomic number. Protons and neutrons are found in the nucleus and electrons are found in the shells surrounding the nucleus. An atom consists of a nucleus surrounded by shells. Atoms are made up of particles called nucleons (protons and neutrons). An atom is the smallest part of an element that can take part in a chemical reaction. The table below shows some elements and their symbols.Įach element contains one type of atom. Different elements are represented by different chemical symbols. They exist as diatomic molecules.Įlements are chemical substances which can not be broken down into simpler substances through chemical means. Group 17 elements are called halogens.Transition elements are d-block elements that make at least one ion with partly filled d-orbital.The entropy of elements in a liquid state is more than the elements which are in a solid state. 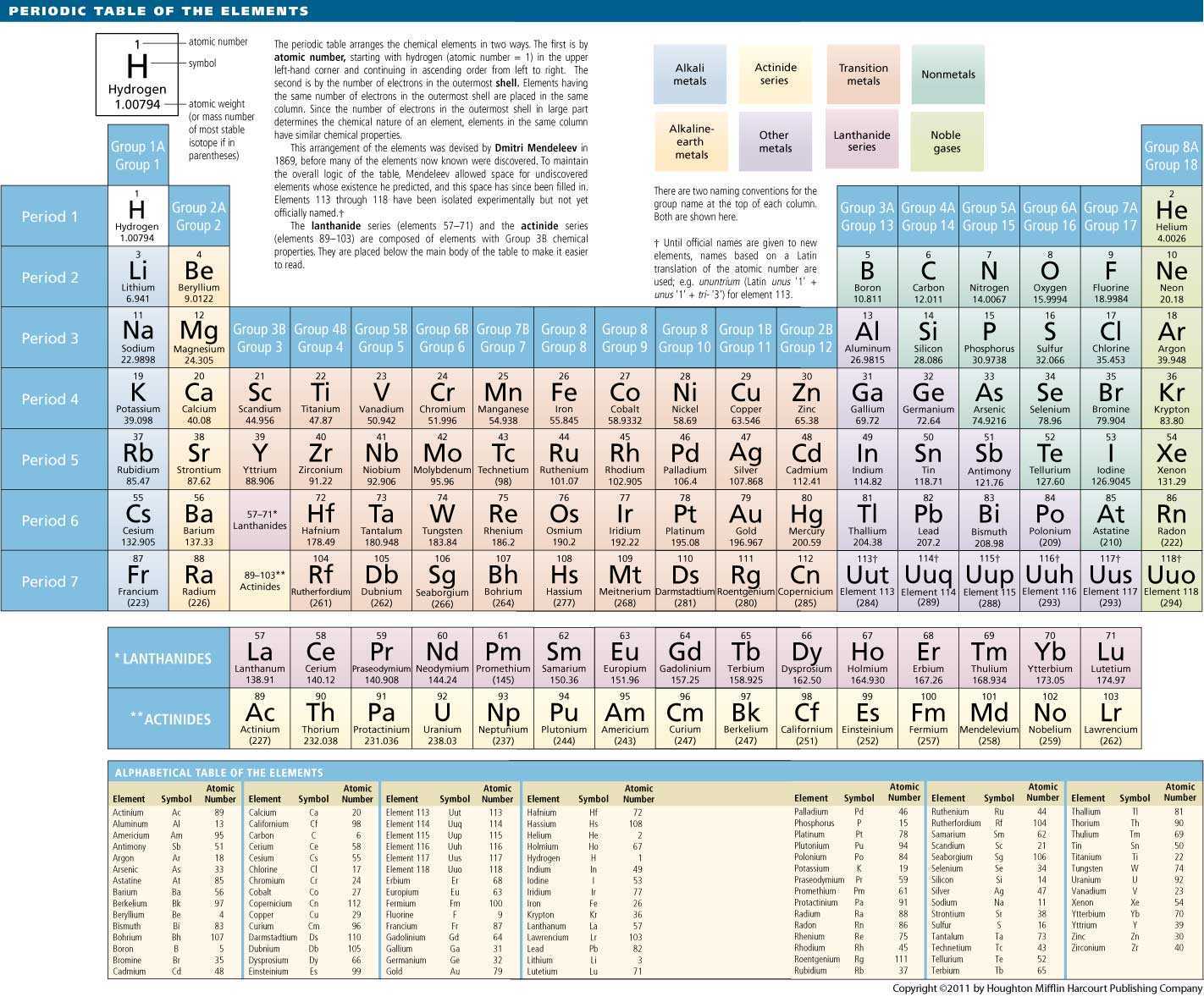
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed


